CIHR Assessment of the Canadian Guideline for Opioids for Chronic Non-Cancer Pain
Table of contents
1. Context
Canada is experiencing a dramatic rise in opioid-related deaths. In 2016 alone, almost 2,500 opioid-related deaths occurred across CanadaFootnote 1. Likewise, the number of opioid prescriptions filled in Canada each year is increasingFootnote 2. In Ontario alone, the number of opioid prescriptions filled increased by five per cent over three years (2013-14 to 2015-16) while the number of people who filled at least one of these prescriptions remained approximately the sameFootnote 3. Inappropriate opioid prescribing practices is one factor contributing to this growing crisis as cases arise of patients becoming addicted while under the care of a physician.
In 2010, recognizing the need to improve safe opioid prescribing practices, a national group of physicians and researchers developed the Canadian Guideline for Safe and Effective Use of Opioids for Chronic Non-Cancer Pain. This guideline provided recommendations to medical professionals on prescribing opioids safely and effectively. Once the 2010 guideline was published, McMaster University’s Michael G. DeGroote National Pain Centre assumed responsibility for keeping it current by highlighting new evidenceFootnote 4. In the years following, the Government of Canada provided funding, through the Canadian Institutes of Health Research and Health Canada, to McMaster University researchers to update the 2010 guideline with new evidenceFootnote *.
The revised 2017 Canadian Guideline for Opioids for Chronic Non-Cancer Pain (the 2017 Canadian Guideline) was published in the Canadian Medical Association Journal (CMAJ) on May 8, 2017. Following its release, The Globe and Mail reported that McMaster University did not exclude medical experts who had received income from opioid manufacturers from the 2017 Canadian Guideline’s voting panelFootnote 5. In particular, the article noted the potential conflict of interest from one panel member, Dr. Sol SternFootnote 6.
2. Overview of the Assessment
In response to public criticism, the Minister of Health directed CIHR to assess what impact, if any, the potential conflict of interest had on the scientific rigour of the 2017 Canadian Guideline. This review aimed to determine whether the 2017 Canadian Guideline provides unbiased, evidence-based guidance to clinicians on opioid prescribing practices.
The CIHR assessment of the 2017 Canadian Guideline consisted of the following three components:
- a review of the process used by McMaster University to develop the 2017 Canadian Guideline
- a review of the process used by CMAJ to make publication decisions; and
- a comparison between the Guideline and an international equivalent.
The assessment was led by the Scientific Directors of the CIHR Institute of Health Services and Policy Research (Dr. Robyn Tamblyn) and the CIHR Institute of Musculoskeletal Health and Arthritis (Dr. Hani El-Gabalawy). The third task involved two Directors from the United States’ National Institutes of Health (NIH): the Director of the National Institute on Drug Abuse (Dr. Nora Volkow) and the Director of the National Institute of Neurological Disorders and Stroke (Dr. Walter Koroshetz).
Dr. Robyn Tamblyn
Scientific Director, CIHR Institute of Health Services and Policy Research
Dr. Robyn Tamblyn of McGill University was selected because of her extensive experience in health policy research. Dr. Tamblyn's ground-breaking research on educational outcomes have clarified the relationships between health professional training, licensure, and practice and subsequently guided credentialing policies. In addition, her work on prescription drug use, its determinants, and ways to improve drug safety has been internationally recognized.
Dr. Hani El-Gabalawy
Former Scientific Director, CIHR Institute of Musculoskeletal Health and Arthritis
Dr. Hani El-Gabalawy of the University of Manitoba was selected because of his extensive expertise in pain management. Dr. El-Gabalawy, an internationally recognized rheumatologist, is a Professor of Medicine and Immunology and a senior clinician-scientist at the University of Manitoba where he holds the Endowed Rheumatology Research Chair.
Dr. Nora Volkow
Director, NIH National Institute on Drug Abuse
Dr. Nora Volkow was selected because of her role as the head of the United States’ National Institute on Drug Abuse and her extensive research experience. Her work was instrumental in demonstrating that drug addiction is a disease of the human brain. She also documented changes in the dopamine system affecting, among others, the functions of frontal brain regions involved with motivation, drive, and pleasure in addiction.
Dr. Walter Koroshetz
Director, NIH National Institute of Neurological Disorders and Stroke
Dr. Walter Koroshetz was selected because of his role as the head of the United States’ National Institute of Neurological Disorders and Stroke and his role as Chair of the National Institutes of Health Pain Consortium. A major focus of Dr. Koroshetz’ clinical research career was to develop measures in patients that reflect the underlying biology of their conditions. With the Massachusetts General Hospital team, he discovered increased brain lactate in Huntington’s Disease patients using MR spectroscopy. He helped the team to pioneer the use of diffusion/perfusion-weighted MR imaging and CT angiography/perfusion imaging in acute stroke.
Their full biographies are included in Appendix A.
3. Review and Observations
i) Review of the Guideline Development Process
The first element of the CIHR assessment was to review the process McMaster University used to develop the 2017 Canadian Guideline. This review examined what impact, if any, Dr. Stern’s perceived conflict of interest had on the scientific rigour of the 2017 Canadian Guideline.
Dr. Tamblyn and Dr. Michelle Peel, Acting Director General of CIHR’s Science, Knowledge Translation and Ethics branch, held a key informant interview with Dr. Jason Busse of McMaster University and primary editor of the 2017 Canadian Guideline. The objectives of this discussion were to review the overall development process and discuss whether this was sufficient to ensure that the 2017 Canadian Guideline is both evidence-based and free from bias.
The development process
The process to develop the 2017 Canadian Guideline began by synthesizing the recommendations from the 2010 version of the guideline and six other recent guidelinesFootnote 7Footnote 8Footnote 9Footnote 10Footnote 11Footnote 12. The team presented this synthesis at a national stakeholder meeting with representatives from patient advocacy groups, governments, medical regulators, the research community, the medical community, and law enforcement. Meeting attendees identified 24 areas where clinical practice recommendations would be helpful.
Governance structure
The research team established four guideline development groups (Figure 1), including one voting panel:
- A Guideline Steering Committee (four members) that led the development process
- A Clinical Advisory Committee (thirteen members) that has clinician experience and a range of perspectives on opioid use for chronic pain
- A Patient Advisory Committee (sixteen members) that informed a statement on patient values and preferences
- A Guideline Panel (a fifteen member panel of clinicians and patients) that developed and voted on the recommendations
Figure 1: Process for Generating the 2017 Canadian Guideline for Opioids for Chronic Non-Cancer Pain
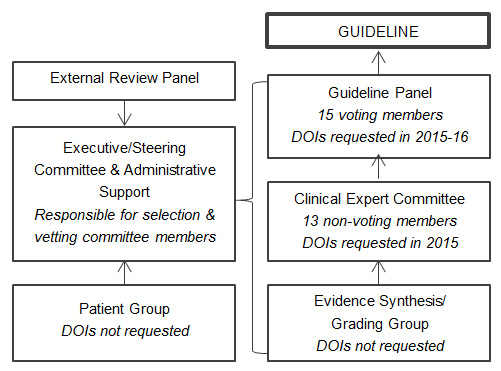
Figure 1 long description
The process for generating the 2017 Canadian Guideline for Opioids for Chronic Non-Cancer Pain was led by the four-person Guideline Steering Committee. This committee was supported by three other groups: 1) a 16-member patient advisory committee that provided advice on patient values and preferences, 2) a 13-member clinical advisory committee that provided advice on opioid use for chronic pain, and 3) an evidence synthese/grading group. Finally, there was a 15-member voting panel comprised of clinicians and patients that voted on the final recommendations that made up the 2017 Canadian Guideline.
To inform the design of the 2017 Canadian Guideline development process, the research team recruited an experienced guideline methodologist, Dr. Gordon Guyatt of McMaster University. Dr. Guyatt, who became the Chair of the Guideline Steering Committee, had been significantly involved in many iterations of the American College of Chest Physicians Evidence-Based Clinical Practice Guidelines on Antithrombotic Therapy and Prevention of Thrombosis, most recently as chair of the ninth edition. The design of the development process was also informed by Clinical Practice Guidelines We Can Trust which was published by the Institute of Medicine, a division of the National Academies of Sciences, Engineering and Medicine of the United States.
Making recommendations
In the Guideline Steering Committee’s view, to be useful, clinical practice recommendations need to be informed not only by evidence but also by context, including patient values and preferences. The Guideline Steering Committee therefore developed a values and preferences statement to guide all recommendations.Footnote 13 This statement was based on a systematic review of the literature on patient values and preferences regarding opioids for chronic non-cancer pain and on focus group interviews with the Patient Advisory Committee.
The Guideline Steering Committee also determined that the panel would only make clinical practice recommendations if there was sufficient evidence to inform the recommendations. Systematic reviews of the evidence were conducted and the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system was applied to move evidence to recommendations. This system is an approach used internationally to grade the quality (or certainty) of evidence and strength of recommendations. As a result, the final 2017 Canadian Guideline included three categories of guidance: “recommendations (supported by evidence from randomized controlled trials or observational studies), good practice statements (supported by indirect evidence), and expert guidance (supported by little or no published evidence).”Footnote 14
After the systematic reviews of the literature were complete, the Guideline Panel and the Clinical Expert Committee met to discuss the evidence and clarify issues for which there was no, or limited, research evidence on which the panel could make its recommendations. In a second meeting of only the Guideline Panel, members voted on 10 recommendations on which there was sufficient evidence. Recommendations required 80% consensus amongst panel members in order to avoid the possibility that a few members could have undue influence on the process. If any panel member disagreed they would record a formal dissenting statement; however, no requests for dissenting statements occurred. The Guideline Panel came to an agreement on all 10 recommendations.
These recommendations were posted online for a one-month public review process. The Guideline Steering Committee reviewed over 500 responses and, in response, revised the language of some recommendations. There were, however, no cases where the public feedback changed the direction (for or against) or strength (weak or strong) of any recommendation.
The final recommendations were presented in a summary paper to the CMAJ. The authors replied to reviewer comments and then the manuscript and guidelines were accepted for publication.
Conflict of Interest
The Institute of Medicine recommends that in the process of guideline development, fewer than 50% of voting members should have financial conflicts of interest (COI).Footnote 15 The Guideline Steering Committee set a higher threshold for managing COI for this guideline. The intent was that the voting Guideline Panel would be composed entirely of individuals without any financial or intellectual conflicts. This was largely successful; no members had any declared intellectual COI and only one of fifteen had a material financial COI.
The 2017 Canadian Guideline required input from both patients and clinical experts, but it was recognized that bias would be prevalent among these groups. As such, the Guideline Steering Committee strove to ensure an equal balance in views among members of both the Clinical Expert Committee and the Patient Advisory Committee, but did not allow these groups to vote on the recommendations or to be present during the voting process.
Due to an administrative oversight, there was one individual on the voting Guideline Panel, Dr. Stern, with a material financial COI; he had received payment for providing medical education talks for industry. Though this was declared prior to voting, it was not brought to the attention of the Guideline Steering Committee through the administrative COI process. After the 2017 Canadian Guideline was developed, the Guideline Steering Committee identified the COI when reviewing the COI forms it collected for submission to the CMAJ. According to Dr. Busse, had this been identified earlier, Dr. Stern would have been proposed for the non-voting Clinical Expert Committee and excluded from the voting Guideline Panel.
Once the COI was identified, the Guideline Steering Committee met with the project coordinator who collected and administered the COI forms and reviewed Dr. Stern’s participation in the development process. The Guideline Steering Committee concluded that there was no evidence of bias during Dr. Stern’s participation. It must be noted that the 2017 Canadian Guideline provides recommendations to avoid prescribing opioids and to prescribe at lower thresholds. In addition, 7 of 10 recommendations focus on harm reduction. These recommendations would not generally be seen to favour industry.
CIHR Observations
In reviewing the process McMaster University used to develop the 2017 Canadian Guideline, CIHR did identify a flaw in how conflict of interest was handled. The COI process was weakened by depending on a single individual (i.e., the project coordinator) to identify potential conflicts. Given the process used to develop the 2017 Canadian Guideline and the entirety of the evidence its committees examined, there is no evidence that a COI did or could have had any real influence on the final recommendations. CIHR found that the 2017 Canadian Guideline development process was rigorous, and concluded that Dr. Stern’s conflict did not ultimately impact the 2017 Canadian Guideline’s recommendations.
ii) Internal Review of the CMAJ pre-publication process
The second element of the CIHR assessment was to review the CMAJ process (Figure 2) that led to the publication of 2017 Canadian Guideline in May 2017.
Figure 2: CMAJ Editorial Process
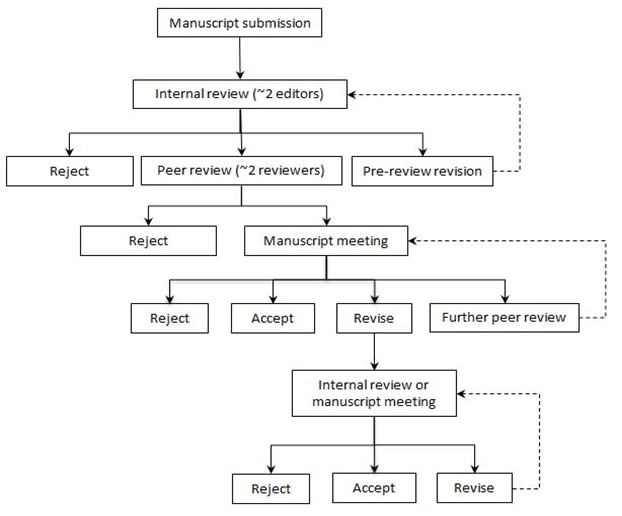
Figure 2 long description
The editorial process for submitting research and guideline articles to the CMAJ is as follows:
- A researcher submits a manuscript
- Editors at the CMAJ do an internal review, which results in either a rejection, revision, or advancing to the peer-review stage.
- At the peer review stage, reviewers assess the article, leading to either rejection or a manuscript meeting.
- Following the manuscript meeting, the article is either rejected, accepted, revised, or undergoes further peer review.
- The last stage is internal review or a manuscript meeting where, after further scrutiny, the article is either rejected, accepted, or revised for the final time.
As part of its review, a senior CIHR official interviewed Dr. Diane Kelsall, Interim Editor-in-Chief of CMAJ, on the journal’s review of conflicts of interest. Prior to accepting the 2017 Canadian Guideline for publication, CMAJ requested authors to complete an International Committee of Medical Journal Editors (ICMJE) form for Disclosure of Potential Conflicts of Interest to disclose any perceived or real conflicts of interest.
The CMAJ reviewed the completed ICMJE forms and acknowledged that perceived or real conflicts existed. Nevertheless, CMAJ felt it was an important guideline to publish. It proceeded to publish the 2017 Canadian Guideline and included the disclosures in the publication.
CIHR Observations
While the conflict of interest processes used by CMAJ and McMaster University were similar, the CMAJ process did use a longer time period to capture self-declared potential conflicts. However, since Dr. Stern declared the potential conflict on both the CMAJ and McMaster University forms, CIHR does not feel the decision to use one form over another had any bearing on the scientific rigour of the 2017 Canadian Guideline.
Following its review, CIHR is confident in the CMAJ process that led to the publication of the 2017 Canadian Guideline. The process identified potential conflicts of interest, considered their weight in deciding to publish, and disclosed them in the publication. This process met international standards for assessing the scientific merit of a publication in a peer-reviewed journal.
iii) External assessment of the Scientific Merit of the 2017 Canadian Guideline
The third element of the CIHR assessment was an independent comparative analysis between the 2017 Canadian Guideline and an internationally-recognized guideline on the same topic. CIHR invited Dr. Nora Volkow, Director of the U.S. NIH National Institute on Drug Abuse, to provide an expert opinion on how the 2017 Canadian Guideline recommendations compare to those developed by the Centre for Disease Control (CDC) in the United States. The CDC Guideline for Prescribing Opioids for Chronic PainFootnote 16 was developed in 2016 to provide recommendations for prescribing opioid pain medication in primary care settings. The CDC Guideline has been endorsed by the U.S. Food and Drug Administration and a number of Canadian colleges of physicians and surgeons (e.g., AlbertaFootnote 17, British ColumbiaFootnote 18, Nova ScotiaFootnote 19). Like the 2017 Canadian Guideline, the CDC Guideline provides recommendations on using opioids to manage non-cancer pain in adults (18 years of age or older) that has persisted for three months or more.
Under Dr. Volkow’s leadership, experts from the National Institute on Drug Abuse (NIDA) and the National Institute on Neurological Disorders and Stroke (NINDS) reviewed the 2017 Canadian Guideline, compared it to the CDC Guideline, and provided some feedback on the 2017 Canadian Guideline for consideration (included in Appendix B). The experts found that the methodology and extent of evidence reviewed to develop both guidelines were similar. In each case, development teams conducted systematic reviews and used standardized categorization of the strength of recommendations. The experts also noted that while some differences exist, the similarities indicate a rigorous development process. The 2017 Canadian Guideline is consistent with available evidence and can be used as a resource in clinical practice.
In addition to the similarities in the development process, the experts identified several similarities in the recommendations of the two guidelines. Both guidelines recommend that opioids should not be the first-line therapy. They also recommend the use of the lowest effective dosage to initiate opioids and careful titration. In addition, the guidelines both further suggest limits on daily morphine of 90 milligram equivalents with special consideration for using larger amounts. Finally, both guidelines recommend co-prescribing naloxone with at-risk patients and that opioids should be tapered and discontinued if they are not effective.
The experts noted only two specific differences. First, that the 2017 Canadian Guideline suggests that controlled release opioids for continuous pain while the CDC Guideline recommends avoiding their use overall and is against their use to initiate opioid treatment. Secondly, the 2017 Canadian Guideline strongly recommends against the use of opioids in patients with active substance use disorder (SUD) and suggests using a non-opioid therapy rather than a trial of opioids. The CDC Guideline, however, does not recommend against the use of opioids in patients with active or history of SUD. Instead, it outlines risk assessment for SUD and recommends providing medication-assisted treatment for those identified with SUD (e.g., co-prescribing naloxone).
The NIDA and the NINDS are of the view that the 2017 Canadian Guideline is based on a rigorous developmental process similar to that used by the Centers for Disease Control and Prevention’s (CDC). Notwithstanding certain differences noted and commentary provided for future consideration, the 2017 Canadian Guideline is consistent with available evidence for clinical practice.
CIHR Observations
In reviewing the comparative analysis submitted by Dr. Volkow, CIHR noted the similarities in the development processes of the two guidelines. It further observed the parallels in the broad strokes of the two guidelines, particularly as related to a preference for non-pharmacologic therapy and non-opioid pharmacologic therapy, daily dosage caps, tapering and co-prescribing naloxone.
Despite the differences mentioned above, CIHR does not feel these reduce the merit of the 2017 Canadian Guideline as a whole. For example, the 2017 Canadian Guideline’s comment on controlled release opioids is not a GRADE recommendation, but one of several guidance statements that provide direction in areas where little published evidence is available. The 2017 Canadian Guideline authors openly note that “the benefit and safety of controlled release or sustained release over immediate release preparation is not clearly established.”
In addition to the comparative analysis, Dr. Volkow provided additional suggestions to inform future iterations of the 2017 Canadian Guideline (refer to Appendix B). CIHR has taken the liberty of providing these to officials at Health Canada under separate cover.
4. Conclusion
Following the three components of its assessment, CIHR finds that the process used to develop the 2017 Canadian Guideline for Opioids for Chronic Non-Cancer Pain was scientifically rigorous despite the flaw in promptly identifying the COI in a single voting member of the fifteen member panel.
Prior to publication, the 2017 Canadian Guideline was subject to many checks and balances in order to minimize undue influence on the final recommendation. These checks included the multi-step process for drafting recommendations, the consensus-based voting process, and the rigorous process to apply for the journal. Coupled with the comparison with the CDC Guideline, it seems clears that the perceived COI of one individual on the voting Guideline Panel did not have any impact on the final recommendations. CIHR concludes that the 2017 Canadian Guideline does provide unbiased, evidence-based guidance to clinicians on opioid prescribing practice that is aligned with international comparators.
Appendices
Appendix A – Biographies
-
Robyn Tamblyn
Scientific Director, CIHR Institute of Health Services and Policy Research
Professor, Department of Medicine and Department of Epidemiology and Biostatistics, McGill University, Faculty of MedicineDr. Robyn Tamblyn is a Professor in the Department of Medicine and the Department of Epidemiology and Biostatistics at McGill University. She is a James McGill Chair, a Medical Scientist at the McGill University Health Center Research Institute, and the Scientific Director of the Clinical and Health Informatics Research Group at McGill University. Dr Tamblyn's ground-breaking research on educational outcomes has elucidated important relationships between health professional training, licensure and practice that have subsequently guided credentialing policies.
Her work on prescription drug use, its determinants, and computerized interventions to improve drug safety (MOXXI) have been recognized internationally. She leads a CIHR-funded team to investigate the use of e-health technologies to support integrated care for chronic disease, and co-leads a Canadian Foundation for Innovation Informatics Laboratory to create advanced technologies to monitor adverse events in populations and create new tools to improve the safety and effectiveness of health care. Her work is published in the Journal of the American Medical Association, the Annals of Internal Medicine, the British Medical Journal, Medical Care, and Health Services Research among others. She has been awarded the CHSRF KT award for her research in improving the use of medication as well as the ACFAS Bombardier award for innovation in the development of a computerized drug management system. As of January, 2011, she became the Scientific Director of the Institute of Health Services and Policy Research at the Canadian Institutes of Health Research.
-
Hani El-Gabalawy
Scientific Director, CIHR Institute of Musculoskeletal Health and Arthritis
Professor of Medicine and Immunology, University of ManitobaDr. El-Gabalawy, an internationally recognized rheumatologist, is Professor of Medicine and Immunology, and senior clinician scientist at the University of Manitoba. He also holds the Endowed Rheumatology Research Chair at that institution. He holds a medical degree from the University of Calgary and has completed postdoctoral fellowships in internal medicine and rheumatology at McGill University.
His research interests have focused on the mechanisms initiating and sustaining joint inflammation in rheumatoid arthritis (RA). Between 1997-2000, as a visiting scientist at the National Institutes of Health, Bethesda, MD, he and his colleagues studied a large cohort of patients with early inflammatory arthritis in order to determine the pathological features seen in the inflamed joints, and to better understand prognosis. A number of seminal observations were made from these studies that have helped clinicians and researchers focus investigative and therapeutic strategies on the early stages of joint inflammation, before permanent progressive damage occurs. Dr. El-Gabalawy has published landmark studies on synovial biology, the pathogenesis of early arthritis, and has recently established a unique First Nations cohort to study gene-environment interactions in the pre-clinical phase of arthritis.
Dr. El-Gabalawy has held a number of administrative positions at the University of Manitoba, including serving as an academic Division Head of Rheumatology and Arthritis Centre Director for three terms, Clinical Director of the Rheumatology Research Laboratory, Residency Program Director, and Chair of the Grants and Allocations Committee for the Health Sciences Centre Foundation in Winnipeg. He has served as a member and Chair of CIHR peer review committees, and has held key leadership positions in The Arthritis Society, the Canadian Arthritis Network Centres of Excellence, and the Royal College of Physicians and Surgeons of Canada. He is also a former member of the CIHR-IMHA Institute Advisory Board.
Over the span of his career, Dr. El-Gabalawy has published more than 100 peer reviewed papers, as well as multiple book chapters and editorials. He has received academic awards and recognitions including an NIH Bench to Bedside Award, fellowship in the Canadian Academy of Health Sciences, and a Queen Elizabeth II Diamond Jubilee Award for service to the arthritis community.
-
Nora Volkow
Director, NIH National Institute on Drug Abuse
Nora D. Volkow, M.D., became Director of the National Institute on Drug Abuse (NIDA) at the National Institutes of Health in May 2003. NIDA supports most of the world’s research on the health aspects of drug abuse and addiction.
Dr. Volkow’s work has been instrumental in demonstrating that drug addiction is a disease of the human brain. As a research psychiatrist and scientist, Dr. Volkow pioneered the use of brain imaging to investigate the toxic effects and addictive properties of abusable drugs. Her studies have documented changes in the dopamine system affecting, among others, the functions of frontal brain regions involved with motivation, drive, and pleasure in addiction. She has also made important contributions to the neurobiology of obesity, ADHD, and aging.
Dr. Volkow was born in Mexico, attended the Modern American School, and earned her medical degree from the National University of Mexico in Mexico City, where she received the Robins award for best medical student of her generation. Her psychiatric residency was at New York University, where she earned the Laughlin Fellowship Award as one of the 10 Outstanding Psychiatric Residents in the USA.
Dr. Volkow spent most of her professional career at the Department of Energy’s Brookhaven National Laboratory (BNL) in Upton, New York, where she held several leadership positions including Director of Nuclear Medicine, Chairman of the Medical Department, and Associate Director for Life Sciences. In addition, Dr. Volkow was a Professor in the Department of Psychiatry and Associate Dean of the Medical School at the State University of New York (SUNY)-Stony Brook.
Dr. Volkow has published more than 600 peer-reviewed articles and written more than 95 book chapters and non-peer-reviewed manuscripts, and has also edited three books on neuroimaging for mental and addictive disorders.
During her professional career, Dr. Volkow has been the recipient of multiple awards. In 2013, she was a Samuel J. Heyman Service to America Medal (Sammies) finalist; and she was inducted into the Children and Adults with Attention-Deficit/Hyperactivity Disorder (CHADD) Hall of Fame. She was elected to membership in the Institute of Medicine in the National Academy of Sciences and received the International Prize from the French Institute of Health and Medical Research for her pioneering work in brain imaging and addiction science. She has been named one of Time magazine’s “Top 100 People Who Shape Our World,” “One of the 20 People to Watch” by Newsweek magazine, Washingtonian magazine’s “100 Most Powerful Women” and “Innovator of the Year” by U.S. News & World Report. Dr. Volkow was the subject of a 2012 profile piece by CBS’s 60 Minutes and was a featured speaker at TEDMED 2014.
-
Walter Koroshetz
Director, NIH National Institute of Neurological Disorders and Stroke
Walter J. Koroshetz, M.D., was selected Director of NINDS on June 11, 2015. Dr. Koroshetz joined NINDS in 2007 as Deputy Director, and he served as Acting Director from October 2014 through June 2015. Previously, he served as Deputy Director of NINDS under Dr. Story Landis. Together, they directed program planning and budgeting, and oversaw the scientific and administrative functions of the Institute. He has held leadership roles in a number of NIH and NINDS programs including the NIH’s BRAIN Initiative, the Traumatic Brain Injury Center collaborative effort between the NIH intramural program and the Uniformed Health Services University, and the multi-year work to develop and establish the NIH Office of Emergency Care Research to coordinate NIH emergency care research and research training.
Before joining NINDS, Dr. Koroshetz served as Vice Chair of the neurology service and Director of stroke and neurointensive care services at Massachusetts General Hospital (MGH). He was a professor of neurology at Harvard Medical School (HMS) and led neurology resident training at MGH between 1990 and 2007. Over that same period, he co-directed the HMS Neurobiology of Disease course with Drs. Edward Kravitz and Robert H. Brown.
A native of Brooklyn, New York, Dr. Koroshetz graduated from Georgetown University and received his medical degree from the University of Chicago. He trained in internal medicine at the University of Chicago and Massachusetts General Hospital. Dr. Koroshetz trained in neurology at MGH, after which he did post-doctoral studies in cellular neurophysiology at MGH with Dr. David Corey, and later at the Harvard neurobiology department with Dr. Edward Furshpan, studying mechanisms of excitoxicity and neuroprotection. He joined the neurology staff, first in the Huntington’s Disease (HD) unit, followed by the stroke and neurointensive care service. A major focus of his clinical research career was to develop measures in patients that reflect the underlying biology of their conditions. With the MGH team he discovered increased brain lactate in HD patients using MR spectroscopy. He helped the team to pioneer the use of diffusion/perfusion-weighted MR imaging and CT angiography/perfusion imaging in acute stroke.
Active in the American Academy of Neurology (AAN), Dr. Koroshetz chaired the professional organization’s Public Information Committee, led the AAN’s efforts to establish acute stroke therapy in the US, founded the Stroke Systems Working Group, and was a member of the AAN Board of Directors.
The four experts declared not having any competing interests in their participation in this review.
Appendix B – National Institutes of Health (US) Review of Canadian Guideline for Opioids for Non-Cancer Pain (August 10, 2017)
Summary
Experts from the National Institute on Drug Abuse (NIDA) and the National Institute on Neurological Disorders and Stroke (NINDS) reviewed the draft [2017] Canadian Guideline for Opioids for Non-Cancer Pain, conducted a comparison with those developed by the Centers for Disease Control and Prevention (CDC), and provided recommendations for consideration. Overall, the draft guideline is based on a rigorous developmental process utilizing a systematic review and standardized categorization of recommendations similar to that used by the CDC. Notwithstanding certain differences noted and recommendations provided for future consideration, the [2017] Canadian Guideline is consistent with available evidence for clinical practice.
Comparison with CDC guidelines
Both sets of guidelines are focused on chronic non-cancer pain and non-end of life pain in adults. Both recommend non-opioid, non-pharmacological approaches prior to initiating opioids for chronic pain in opioid naïve patients, and in combination with patients who are prescribed opioids. The methodology and expanse of evidence reviews were similar with systematic reviews conducted and standardized categorization of the “strength” of recommendations.
- [The 2017 Canadian Guideline] provides detailed evidence-based information for clinical/patient decision points for prevalent pain conditions or clinical scenarios, such as history of substance abuse disorder, specific to each recommendation.
- [The 2017 Canadian Guideline] provides details and comparative evidence on efficacy and side effects for opioids with other current therapies for prevalent pain conditions, and for comparative evidence for different opioid based approaches in each recommendation.
- [The 2017 Canadian Guideline] includes evidence based guidance on related health issues (e.g. immediate vs controlled release opioids, co-prescribing cautions, opioids and sleep apnea, secondary hypogonadism, risk mitigation, fentanyl, naloxone co-prescribing).
- The CDC Guideline includes tables that can be applied generally to inform all recommendations.
- Table on comparative evidence for specific treatment modalities and the pain conditions that were studied.
- Table on evidence for risk and adverse effects of opioids.
- The CDC Guideline provides detailed guidance for providers to communicate with patients on setting treatment plans, goals, expectations, and risks.
- The CDC provides guidance to prescribers to identify risks for opioid harms (urine testing, PDMP use, co-prescribing, misuse, etc.).
- The CDC provides detailed guidance on frequency of follow-up to assess risks benefit balance to guide the treatment plan.
Specific differences
- Canadian recommendations suggest use of controlled release opioids for continuous pain; CDC recommends avoiding use of controlled release formulations overall and recommends against their use to initiate opioid treatment.
- [The 2017 Canadian Guideline] strongly recommends against the use of opioids in patients with active substance abuse disorder and weakly recommends non-opioid rather than opioids for patients with past history of substance abuse disorder; the CDC does NOT recommend against use of opioid in patients with active or history of SUD and outlines risk assessment for SUD, and recommends providing MAT for those identified with SUD.
Specific similarities
- Caps on MME’s (initiate at 50, cap at 90- with special consideration for over 90)
- Use of lowest effective dosage to initiate opioids, careful titration, tapering when appropriate
- Co-prescribing naloxone with at risk patients
Feedback for Consideration
- The CDC Guideline recommends that opioid therapy should be complemented with non-opioid therapy when used. Consider adding this recommendation for a multidisciplinary approach to pain treatment.
- Both the CDC Guideline and the [2017 Canadian Guideline] recommend that opioids should not be first-line therapy; the [2017 Canadian Guideline] states that opioid therapy should be prescribed if non-opioid therapies fail (the CDC Guideline does not directly encourage this). Consider adding more detailed guidance on what types of persistent problematic pain opioids are likely to be effective in treating.
- Both the CDC Guideline and the [2017 Canadian Guideline] recommend that opioids should be tapered and discontinued if they are not effective; the CDC Guideline bases tapering and discontinuation on a benefit/harm ratio, while the [2017 Canadian Guideline] bases this on improvement in pain or function. Consider including a discussion of the risk/benefit considerations.
- Consider providing more discussion on use of immediate release vs. extended release/long- acting opioids upon initiation.
- Consider providing additional guidance on developing treatment goals and communication strategies for discussing risks/benefits and treatment expectations with patients.
- Consider including a more detailed guidance to prescribers to identify risks for opioid harms (urine testing, PDMP use, co-prescribing, misuse, etc.) as well as guidance on frequency of follow-up to assess risks benefit balance to guide the treatment plan.
- For recommendation 9: Consider more explicitly discussing the impact of withdrawal when tapering and its impact on patient perceptions of treatment outcomes; also, consider discussing screening for illicit opioid use when tapering patients.
- Recommendation 10: when discussing pain the document notes that you were uncertain about the effects of multidisciplinary programs on pain. It should be made clear that this is specifically referring to when patients are tapered off high-dose opioids.
- The best practice statements on page 77 (monitoring) recommends monitoring for response to treatment. Consider also recommending monitoring for opioid misuse and addiction.
Format and Organization
- Page 16: This sentence is unclear: “Carbamazepine may have a higher effectiveness relative to opioids (tramadol) as has another anticonvulsant (gabapentin), and the antidepressant amitriptyline may have lower mean costs and higher effectiveness than tramadol. [36]
- The boxes on preferences and values states, “Patients with chronic non-cancer pain may place little importance on avoiding rare but serious side effects such as addiction, overdose, or death, and are often willing to trade the risk of these effects for small but important pain relief.”
- This seems like an overgeneralization. Many patients have significant concerns about these issues. Perhaps qualify by saying “some patients”.
- Page 72 states “The potential harms of opioids generally increase with dose, and some may not be attributed to the drugs (particularly depression, hormonal disturbance, sleep disturbance and opioid-induced hyperalgesia).
- Opioid-induced hyperalgesia would by definition be attributed to the drug.
- As noted in the document, organization and readability can have a significant impact on uptake. It may be helpful to reorganize the flow of the document focusing on concise presentation of the most salient recommendations. For example, for each for each scenario include the recommendations for what to do, what not to do, what to monitor for, etc.
- Date modified: